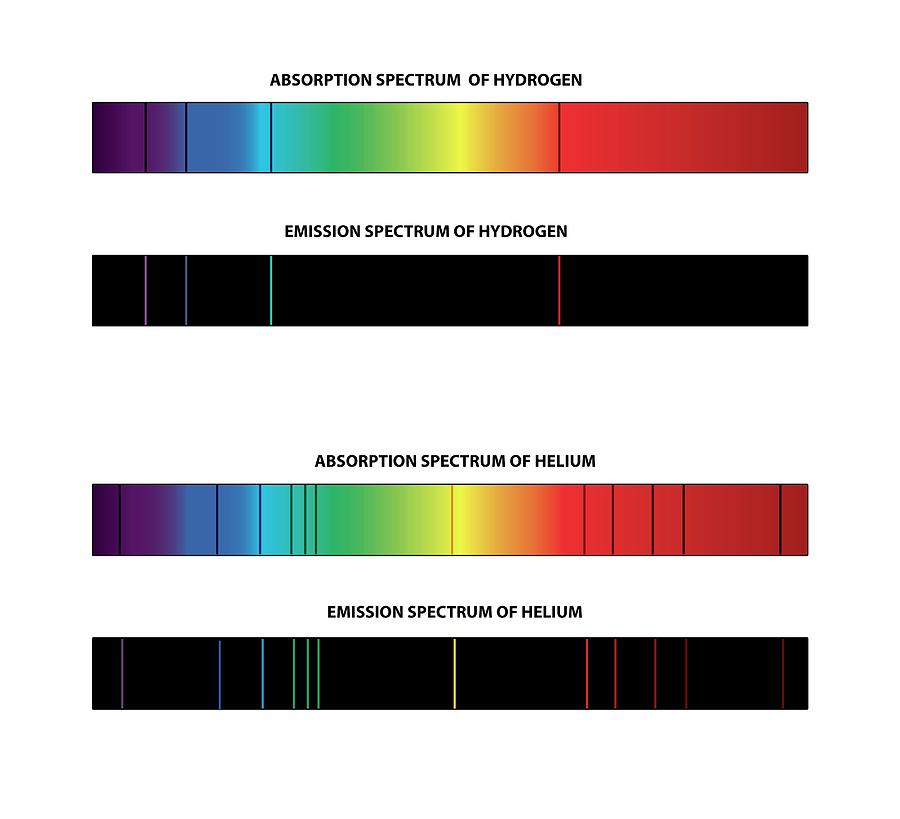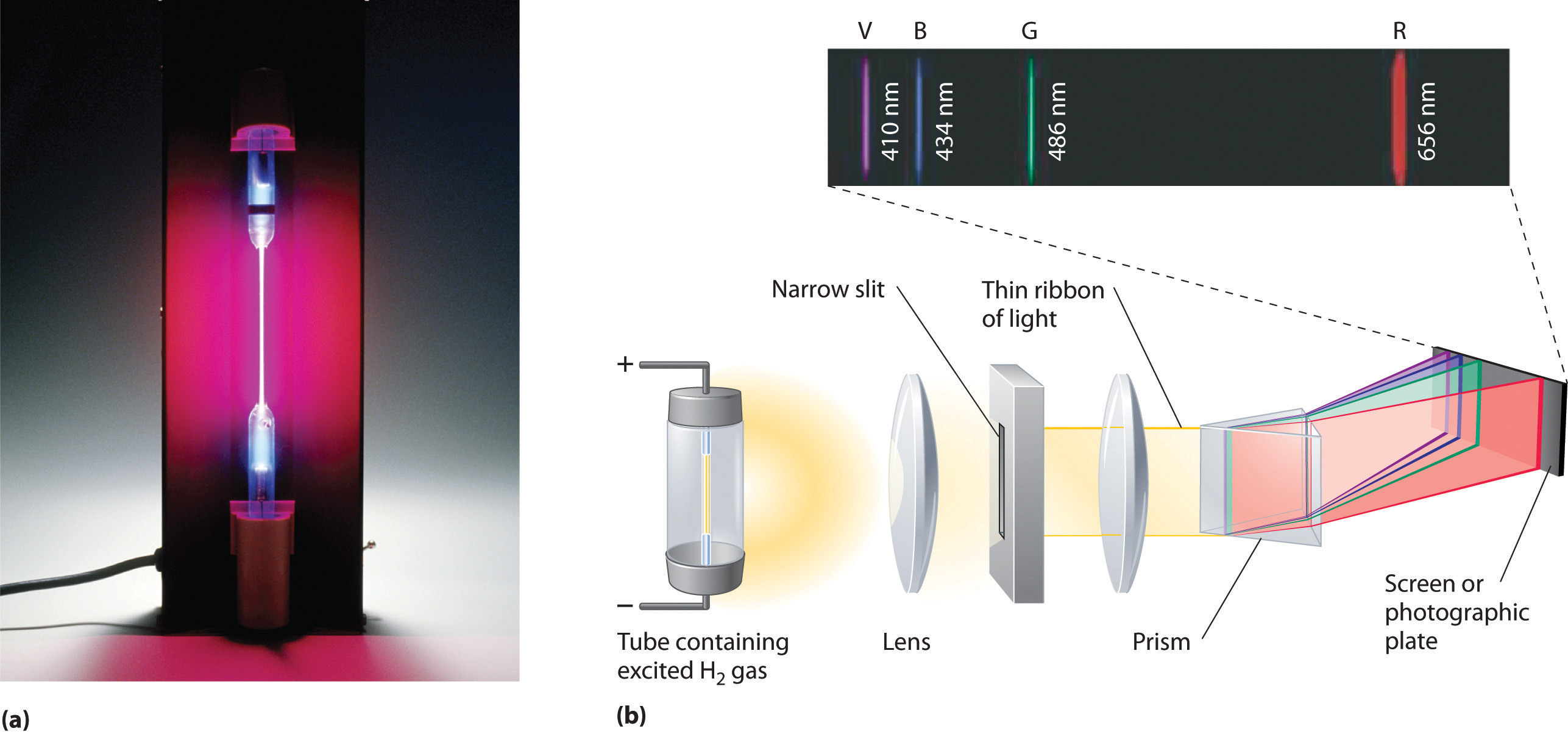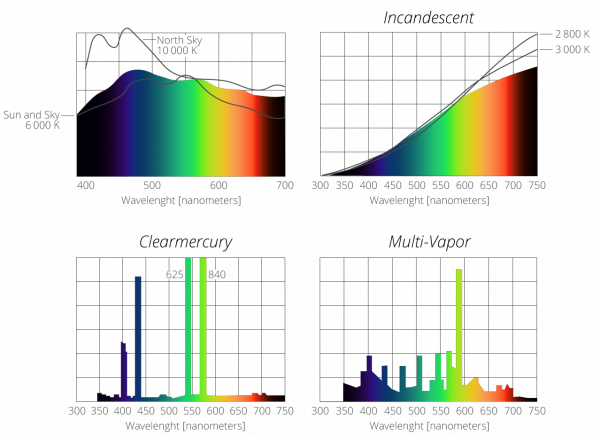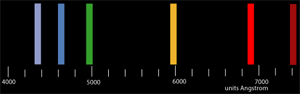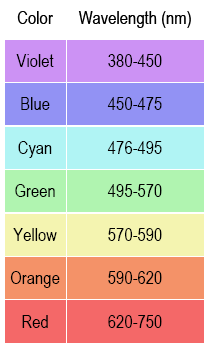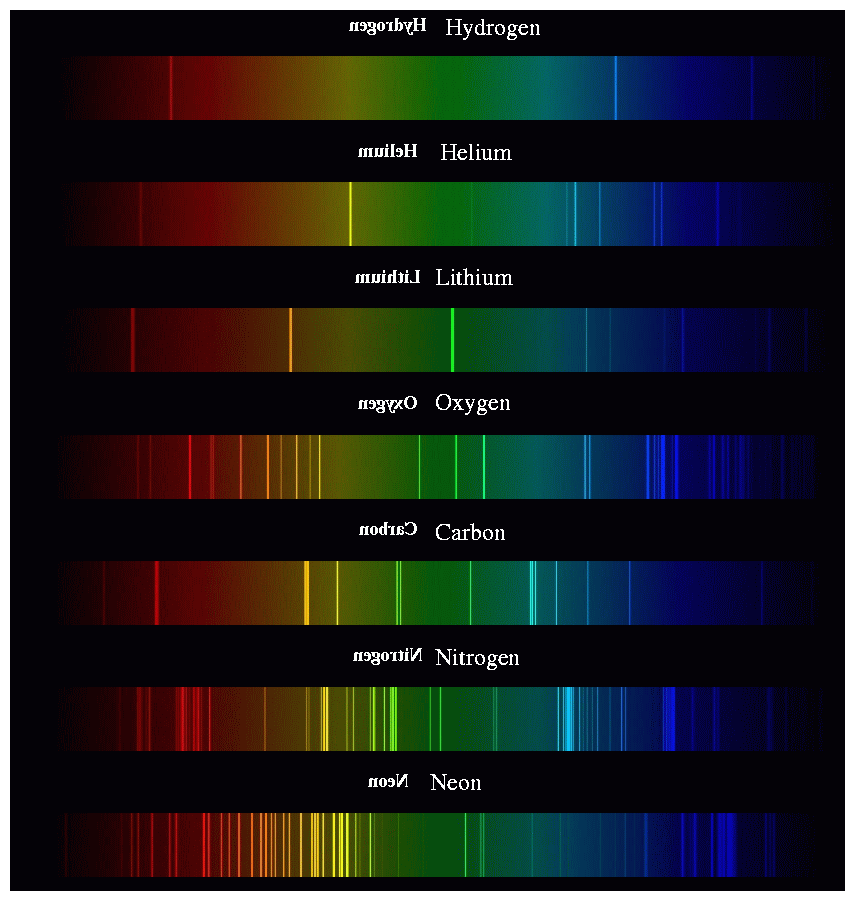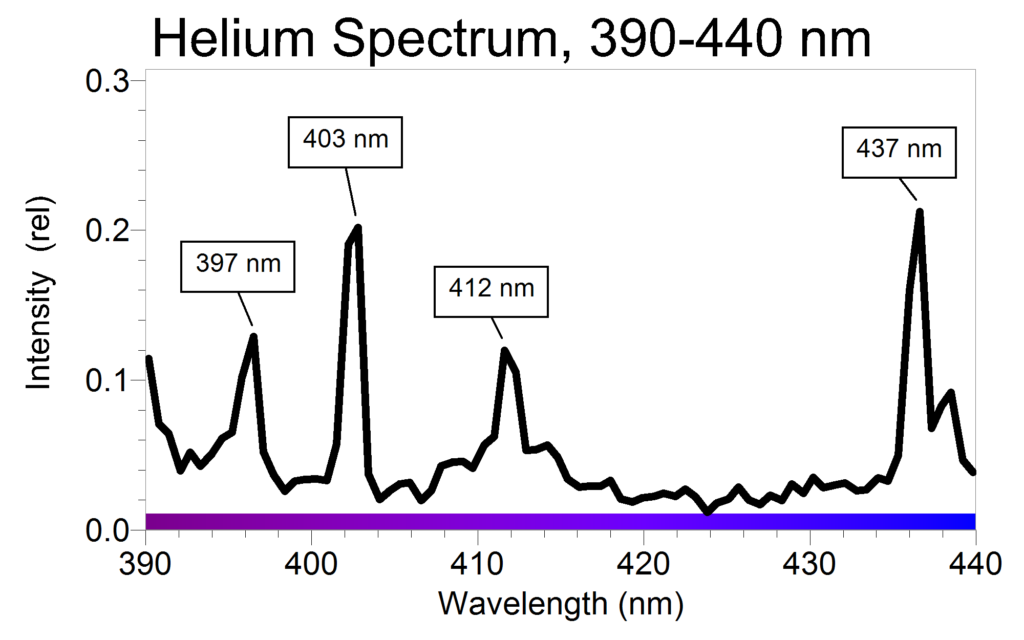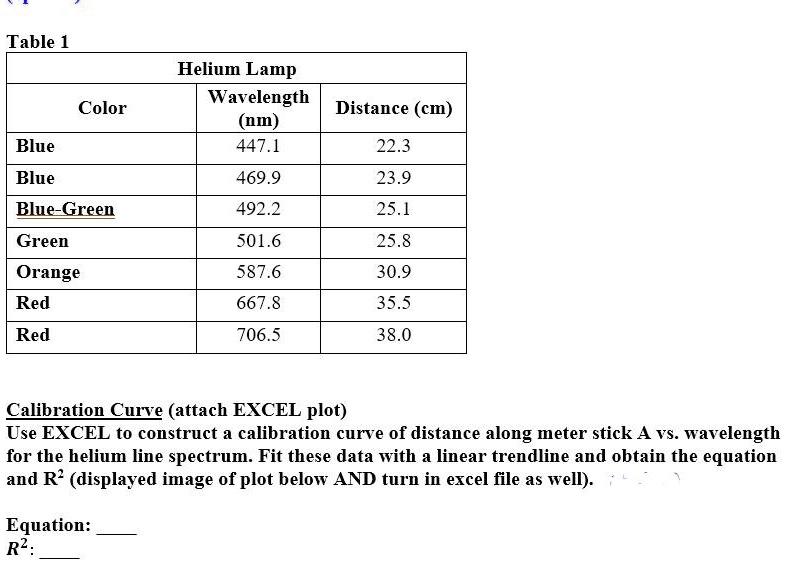
SOLVED: Table Helium Lamp Wavelength Distance (cm) (nm) 447.1 22.3 Color Blue Blue 469.9 23.9 Blue-Green 492.2 25.1 Green Orange Red 501.6 587.6 25.8 30.9 667.8 35.5 Red 706.5 38.0 Calibration Cuve (
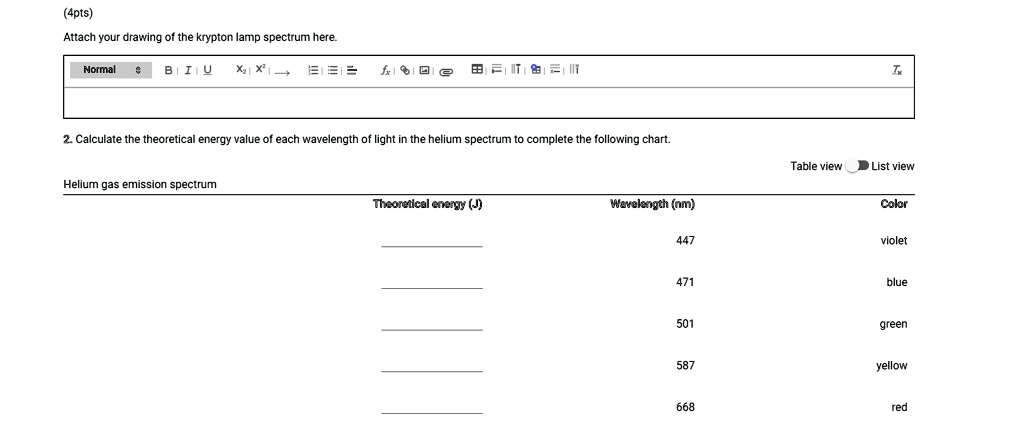
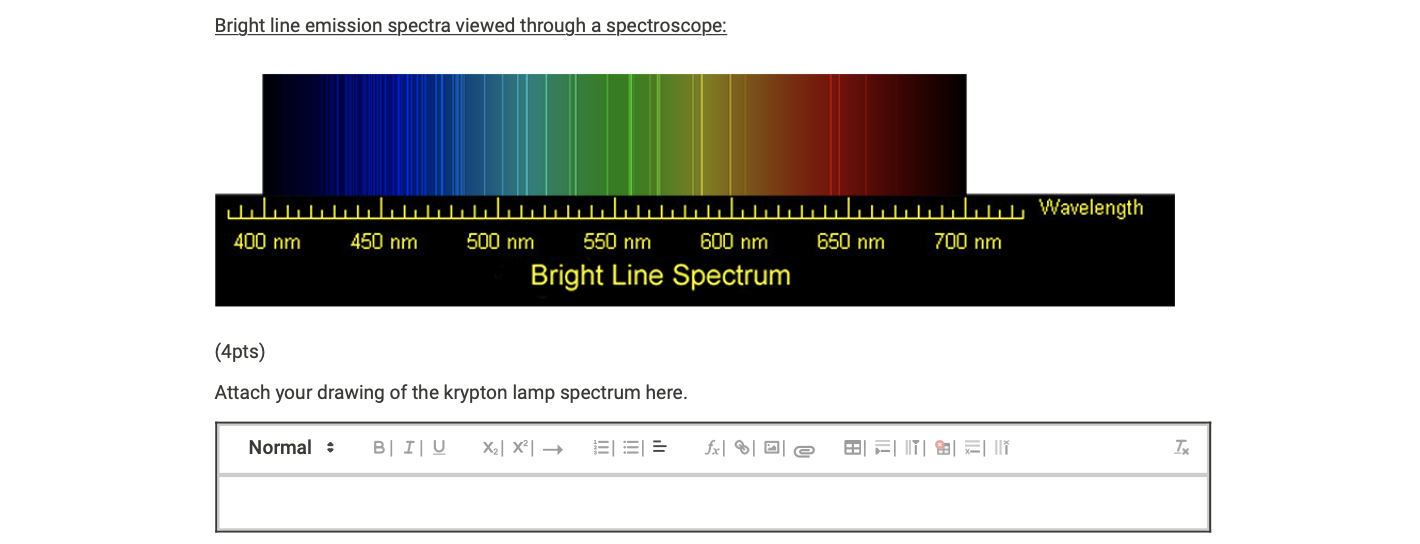
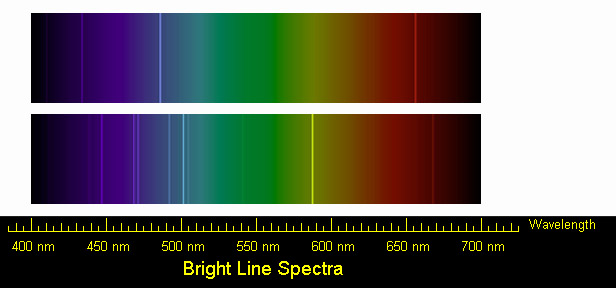
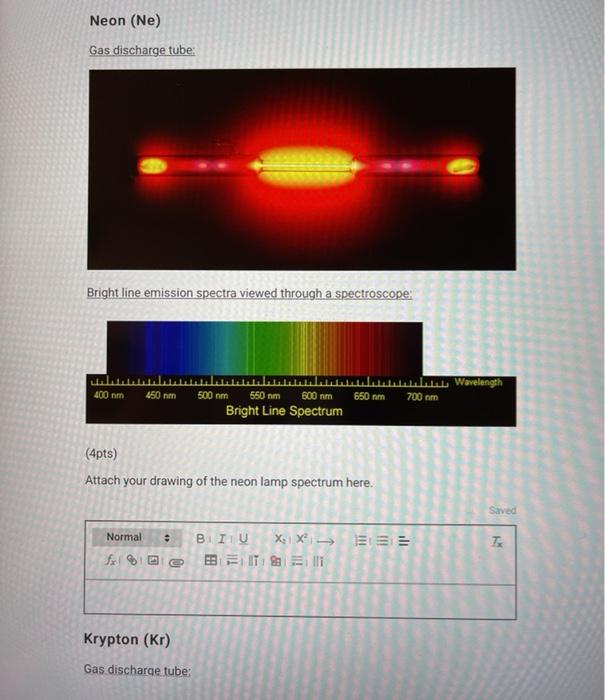
SOLVED: (Apts) Attach your drawing of the krypton lamp spectrum here Normal B / I 4 X1c 031= Jx| %/012 B FM 8 = Ii Calculate the theoretical energy value of each

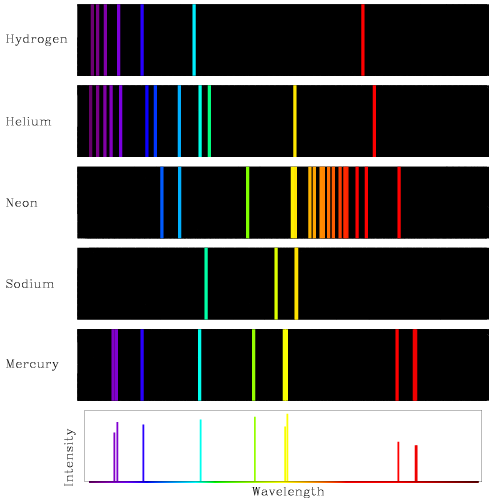
Emission Spectra Gas Lamp (Helium) | Central Coast Science Project | Science Kits for Teaching Library